GMP compliance services for gene and cell-based therapy (ATMPs)
ATMPs
Advanced Therapy Medicinal Products (ATMPs) refer to a new group of medicinal products that use modern genetic engineering. These include cell-based therapy, somatic gene therapy, genome editing, CAR-T cell therapy, single cell analysis and tissue products.
About Advanced Therapy Medicinal Products (ATMPs)
Currently there is a growing focus on advanced therapy medicinal products (ATMPs) as research has made remarkable progress. New possibilities have emerged for the next generation of targeted treatments.
Cell and gene therapies are related but they are not the same. Both are among the precision medicines to directly repair patient-specific DNA mutations.
Today, gene therapy represents the largest group of ATMPs.
ATMP Definitions
Advanced Therapy Medicinal Products (ATMPs), refer to a new group of medicinal products that use modern genetic engineering. These include cell therapy, somatic gene therapy, genome editing, CAR-T cell therapy, single cell analysis and tissue products. Today, gene therapy represents the largest group of advanced therapy medicinal products (ATMPs).
Gene Therapy
Gene therapy is a special form of therapy that treats diseases that are usually caused by a single defective gene. Gene therapy is designed to repair gene defects by replacing or repairing these errors and with the mistake corrected the gene should function properly and the disease should be eliminated or alleviated.
Gene Technology
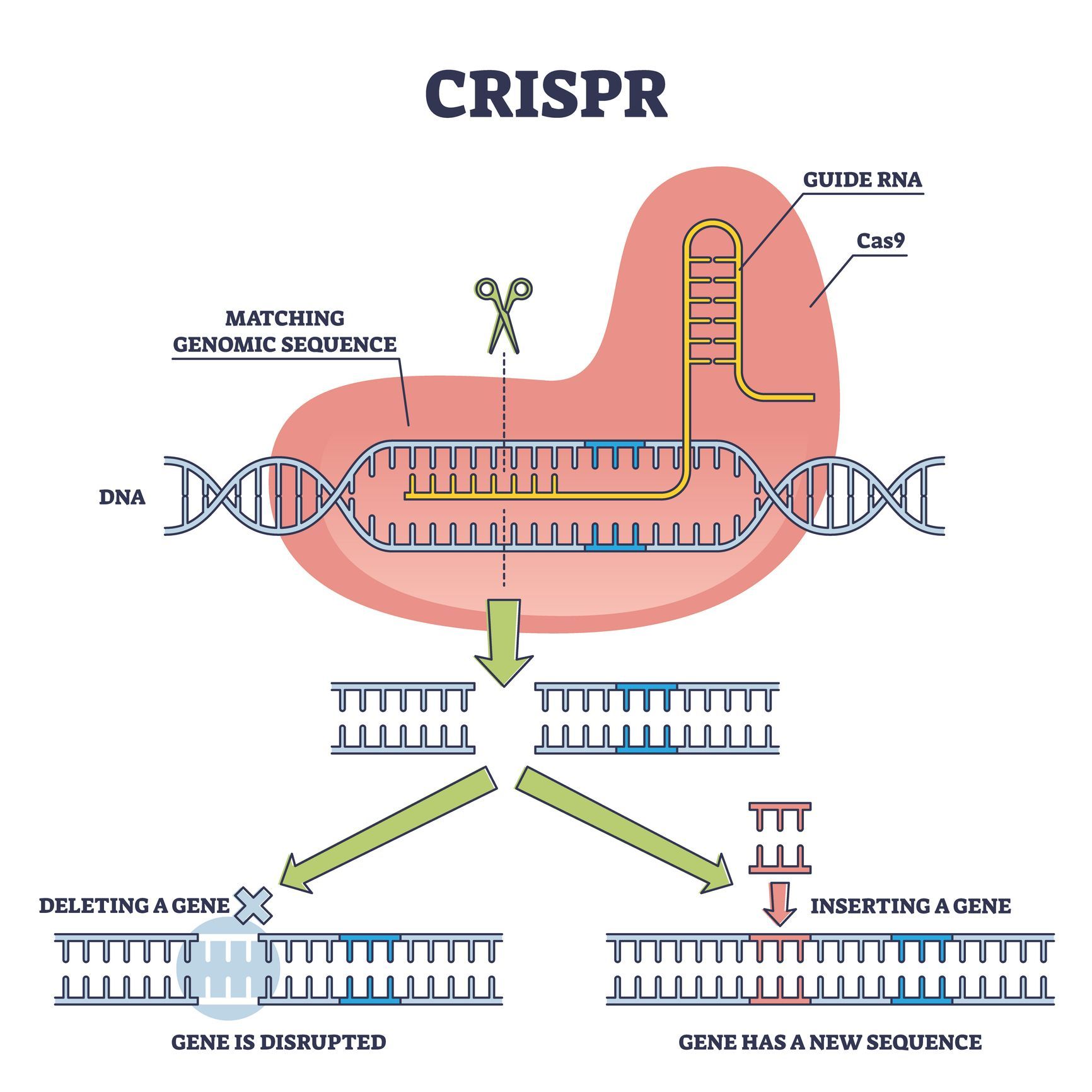
About CRISPR/Cas
Clustered Regularly Interspaced Short Palindromic Repeats
Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) are genetic elements found in the genome of prokaryotics as part of an acquired immune system against bacteriophages.
The CRISPR/Cas system memorizes previous infections by integrating short sequences of the invaded virus genome. Upon reoccurring infections, the CRISPR/Cas recognizes and cleaves the foreign DNA or RNA.
The CRISPR/Cas system is also referred to gene scissors because it can cleave DNA with surgical precision at a specific site in the genome, allowing scientist to switch genes off or insert new sequences at the cutting site.
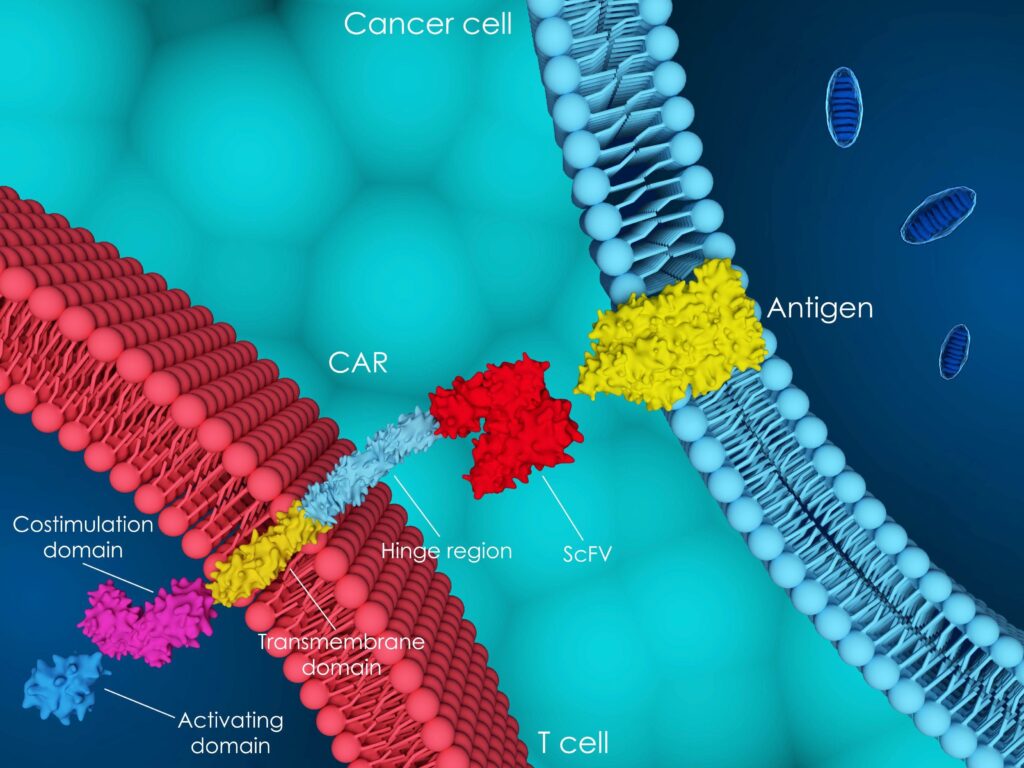
About CAR-T cell therapy
Chimeric Antigen Receptor (CAR) T cell therapy is a form of immunotherapy using T cells collected from the patients for treatment.
The mode of action of CAR-T cell therapy depends on the interaction of Chimeric Antigen Receptor (CAR) in which the CD3 complex is associated.
In using gene technology, the cells are genetically engineered by the treatment centers manufacturer’s laboratories. The T Cell Receptor (TCR) is genetically modified to become a Chimeric Antigen Receptor (CAR).
CAR T cell therapy offers new precision treatment for certain forms of blood disorders. CAR T cell therapy is unique because the cells are individually generated for each patient.

GMP Compliance of AMTPs

Regulation of ATMPs
This regulation lays down the regulatory requirements concerning the authorization, supervision and pharmacovigilance of advanced therapy medicinal products. The approval procedure ensures the quality, efficacy and safety of these medicinal products.
Gene therapies require manufacturers to implement validated processes in order to guarantee safe handling. Article 5 of the Regulation (EC) 1394/2007, states compliance with Good Manufacturing Practices (GMP).
Consistent product quality is mandatory but especially for gene and cell-based therapies arduous and demanding. Compliant GMP manufacturing is hampered by small scale batches and non-routine processes of ATMPs.
Manufacturing Authorization
Nicht routinemäßig hergestellte ATMP nach § 4b AMG
Applications for approval of advanced therapy medicinal products (ATMPs) according to §4b paragraph 3 AMG (Arzneimittelgesetz) based on non-routine production must be submitted to the Paul Ehrlich Institute.
„We are contributing to strategies and validated operations for GMP release of gene and cell-based therapy.“
Key Benefits
We are contributing to scientific and laboratory assistance with genetic engineering, cell culture procedures and quality control operations. We supply hands-on dexterity with cell-based assays, vector engineering (AVV) and cell line engineering.

Our Services

- Contributing to scientific and laboratory assistance
- Facilitating the transition to GMP compliant processes
Your Benefits

- GMP compliant manufacturing of gene and cell-based therapy (ATMPs)
- Manufacturing authorization of ATMPs
- Clinical trial and marketing authorization of ATMPs